pgs. 3A-7.doc
Phase I/II Trial to Document
Immunomodulatory, Cytotoxic and
Potentially Anti-HIV Properties of Iscador
(IND# 32,829)* (Original title: Phase I/II Studie zur Dokumentation der immunmodulierenden, zytotoxischen und der moeglichen anti-HTV-Wirkungen von Iscador (IND # 32,829). Merkurstab 1995; 48: 124-8. English by A. R. Meuss, FIL, MTA.)
Introduction
At the San Francisco General Hospital Aids Clinic, extensive clinical experience
with the use of Iscador for HIV-positive and AIDS patients has been
gained since 1984. The impression has been that Iscador QuFrF (information
supplied by Weleda: Due to legal restrictions, this preparation is not
obtainable in Germany.) shows better anti-HIV activity than Iscador Qu.
With this and with the help of Prof. Maria Linder from the Department of
Biochemistry at Fullerton University, we were able to provide sufficient
clinical and preclinical data to obtain permission to do a trial with Iscador
QuFrF and Iscador Qu Special (Iscador QuS). To our knowledge this was the
first time a purely plant-based preparation achieved the status of
Investigations New Drug (IND) so that the trial could begin.
Having obtained permission of the Food and Drug Administration, a
phase I/II trial with Iscador QuFrF and Iscador QuS was done in Germany. It
was a prospective, longitudinal, open-label, rising dose trial with HIV-
positive (N = 46), healthy, non-smoking subjects (N = 24) and cancer patients
(N = 8). A diurnal profile was additionally produced for 21 Iscador-naive
subjects. The period of observation was 6-15 months in most cases.
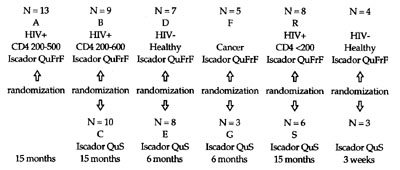
Iscador QuFrF is a special mixture of oak-grown mistletoe extracts, an
experimental preparation of the Hiscia Institute in Arlesheim/ Switzerland
that has proved its value in many individual cases of treating HIV-positive
patients.
Iscador QuS is the usual oak-grown mistletoe preparation produced by
lactic acid fermentation of an aqueous extract, with the total mistletoe lectin
concentration at a strength of 5 mg.
Objectives
The trial was designed to answer the following questions:
- Does exhibition of Iscador QuFrF and Iscador Qu Special in doses of 0.01
mg, 0.1 mg, 1.0 mg, 2.0 mg, 5.0 mg and 0.1 mg/kg of body weight produce
toxic side effects demonstrable by full blood profile, clinical chemistry and
clinical observation of HIV-positive individuals, cancer patients and
healthy subjects?
- Do Iscador QuFrF and Iscador Qu Special have potential anti-HIV
activities?
- Do Iscador QuFrF and Iscador Qu Special have potential immunomodulatory
actions?
- Could Iscador QuFrF and Iscador Qu Special enhance cytotoxic actions
and anticancer activities?
- Is there an optimal posology for Iscador QuFrF and Iscador Qu Special,
determinable by immunomodulatory anti-HIV and cytotoxic parameters?
Methods
HIV-positive subjects were mainly recruited with the help of AIDS-HiIfe in
Cologne, medical practitioners and the AIDS Advisory Center in Duesseldorf
(N = 46; mean age 36 [+ 7]; CD4 < 600; male N = 44; female N = 2; CDC
classification stages n-IVC; Kamofsky index > 70%).
The healthy, non-smoking subjects were mainly medical students,
physicians and other staff at Herdecke Hospital (N = 24; mean age 29 [+ 7];
male N = 15; female N = 9; Kamofsky index > 90%).
Cancer patients were referred by colleagues at Herdecke Hospital (N = 8;
mean age 51 [+ 18]; male N = 0; female N = 8; Kamofsky index > 70%).
The trial was given a positive rating by the Main/Rhein Ethics
Commission in Frankfurt/Main.
Routine laboratory tests such as full blood profile and clinical chemistry
were done at the clinical laboratories at Herdecke Hospital (Chr. Rhem,
Biochem. Dipl.). Immunologic laboratory investigations such as flow
cytometry, ELISA, HIV-p24 antigen and locomotion in 3D collagen matrix
were performed at the Institute of Immunology, Witten/Herdecke
University (Prof. K. Zaenker) and the clinical laboratories of Dr. H. Knechten
in Aachen. Histologic studies were done at the pathological histology
laboratory of Dr. H. Laaff in Freiburg.
Inclusion criteria/or HIV-positive individuals
- HIV-antibodies or HIV-p24 antigen positive
CD4<600
- Age between 18 and 65 years
- No active infection
- Primary or secondary prevention of opportunist infections and AZT, DDI
or DDC medication was permitted if the patient had started on this not less
than 3 months earlier, tolerated it well and intended to continue on the
regimen while participating in the trial.
Inclusion criteria/or healthy, non-smoking subjects
- HIV-antibody negative
- Age between 18 and 65 years
- Non-smoker for not less than 3 months
- Not taking specific hormones liable to influence the immune system (incl.
steroids, contraceptive pill).
Inclusion criteria/or cancer patients
- Malignant condition histologically confirmed
- Stable clinical condition
- Age between 18 and 65 years
- No chemotherapy or hormones for not less than 6 weeks.
Prior to giving Iscador, blood was taken 2 or 3 times at 5-7 days intervals
from all subjects. The mean values of the tests provided a reference baseline
for each individual.
At the beginning of the trial, subjects attended the clinic once a week,
later every 2 weeks. Blood was always taken between 7.00 and 9.30 a.m. to
minimize the effect of circadian variations.
All differential counts were determined twice, each time by the same
technicians at Herdecke Hospital.
Numbers being small in each group and individual groups being non-
homogeneous, it was not possible to establish a normal distribution, and only
non-parametric tests could be used.
In each group and for each parameter, the hypothesis was tested for
unvarying effects with rising doses. The Friedmann test for linked samples
was used; it is the pendant to the parametric F test in variance analysis. The
level of significance chosen was a = 0.05.
The Wilcoxon sign test was used to test the hypothesis of a difference in
the desired parameter between baseline and the different doses. This is based
on the ranks for the difference between baseline and results with 0.01 mg, 0.1
mg, 1.0 mg, 2.0 mg, 5.0 mg and the maximum dose of 0.1 mg/kg body
weight. The a level was again corrected by the Bonferoni method.
Preliminary Results
Both Iscador QuFrF and Iscador Qu Special were well tolerated. No toxic side
effects were noted. The clinical condition of all HTV-positive individuals and,
with one exception, of all cancer patients remained stable or improved. No
patient left the trial because of undesirable side effects or reduction in life
quality. None of the HIV-positive individuals showed deterioration of their
clinical condition during the 15-month period of observation.
The CD4 percentage remained stable in each HIV-positive group, as did
the CD4:CD8 ratio in all patient groups. Reduction in CD4:CD8 was a trend
noted only in the group of healthy, non-smoking subjects. CD3/25 positive
lymphocytes (indicative of IL-2 production) increased between Iscador
QuFrF 0.1 mg and 2.0 mg (p < 0.01) and between Iscador Qu Special 2.0 mg
and 5.0 mg, the rise being significant in 2 groups (p < 0.01).
IL-2 is the most important growth factor for T lymphocytes. Th2
lymphocytes, which do not produce IL-2 but use IL-4 as an autocrine growth
factor, show a higher profile after IL-2 stimulation.1 IL-2 stimulates B
lymphocyte proliferation, immunoglobulin secretion and differentiation
(IgGI and IgM).(2,4) It also stimulates the dominant Th1 response, plays an
important role in inhibiting progression of HIV infection and activates cell-
mediated immunity.(5)
In group A, treated with Iscador QuFrF, serum beta -2 microglobulin
showed a significant decrease at 5.0 mg. This is a positive prognostic sign
with HIV, demonstrating inhibition of progressive HIV infection.
p24 antigen almost always remained negative if it had been negative in
the baseline. In several HIV positive individuals p24 antigen showed a
marked decrease during the trial.
Skin biopsies at the injection site were taken from 7 healthy, non-
smoking subjects. T lymphocyte infiltration was found, something not
previously reported. We hope to give details of this in due course.
At the beginning of the trial, all groups would repeatedly show a
temporary lymphocytopenia which we consider to be a sign of
compartmental shift. Immunomodulated lymphocytes leave the blood
stream and penetrate into tissue.
In a 3-dimensional collagen matrix, we found increased lymphocyte
locomotion ex vivo in vitro for some of the subjects. A 3-year project is in
progress to investigate this phenomenon in vitro and ex vivo in vitro
(compartmental shift: increased locomotion and cytotoxic defense against
cancer cells).
Shortly after the start of the trial we noted significant, lasting eosinophilia
(p < 0.05) in most subjects.
Summary
Preliminary evaluation of this phase I/II trial with Iscador QuFrF and Iscador
QuS for HTV-positive individuals, cancer patients and healthy, non-smoking
subjects shows that both Iscador preparations have immunomodulatory
properties but that the anti-HIV activity of Iscador QuFrF in HIV-positive
individuals appears to be greater than that of Iscador QuS. Conversely,
Iscador QuS appears to give better activation of CDS-positive lymphocytes.
Iscador QuFrF may therefore be important in the treatment of HIV-positive
individuals, whereas Iscador Qu Special is more suitable for the treatment of
cancer patients.
Iscador QuFrF has a distinctly higher total mistletoe lectin concentration
than Iscador QuS, which may explain why maximum IL-2 production was
noted with Iscador QuFrF 1.0 mg to 2.0 mg, and Iscador Qu Special 2.0 mg to
5.0 mg.
Two multicenter trials are planned for Spring 1995 - a phase III trial
(efficacy trial) with Iscador Qu Special for women with cervical dysplasia
(CIN I and II) and a phase II trial with Iscador QuFrF for individuals with
HIV infection. Both trials will be conducted by the Institute of Oncologic and
Immunologic Studies.
Robert Gorter, M.D.
Instistitut fuer onkologische und immunologische Forschung
Krankenhaus Moabit
Turmstr. 21
D-10559 Berlin
Germany
References and Note
1 Smith KA. Interleukin 2: inception, impact and implications. Science 1988; 240:1169-76.
2 Zubler RH, Loewenthal JW, Erad F, Hashimoto N, Devos R, MacDonald HR. Activated B cells express receptors for, and proliferate in response to, pure interleukin 2. / Exp Med 1984; 160: 1170-83.
3 Coffman RL, Seymour BW, Lebman DA et al. The role of helper T cell products in mouse B cell differentiation and isotype regulation, Immunol Rev 1988; 102:5-28.
4 O'Garra A, Murphy K. Role of cytokines determining T lymphocyte function. Current Opinion in Immunology 1994; 6:458-66.
5 Horton R. Renegade HIV immunity hypothesis gains momentum. Lancet 1993; 342:1545.
All calculations were made using the SAS program version 6.04, graphics with Harvard
Graphics, version 3.0.